HPLC / UPLC purity
analysis
API Impurities & Reference Standards
Precision-engineered impurity standards and analytical support to ensure regulatory compliance and product integrity.
Precision-Engineered Impurity Standards for Global Compliance

Elitekem Labs specializes in the design, synthesis, and characterization of API impurities and reference standards, supporting pharmaceutical companies in meeting regulatory expectations and accelerating drug development timelines.
We deliver high-purity impurity standards and custom reference materials tailored to your molecule, process, and submission strategy.
Our Analytical Journey
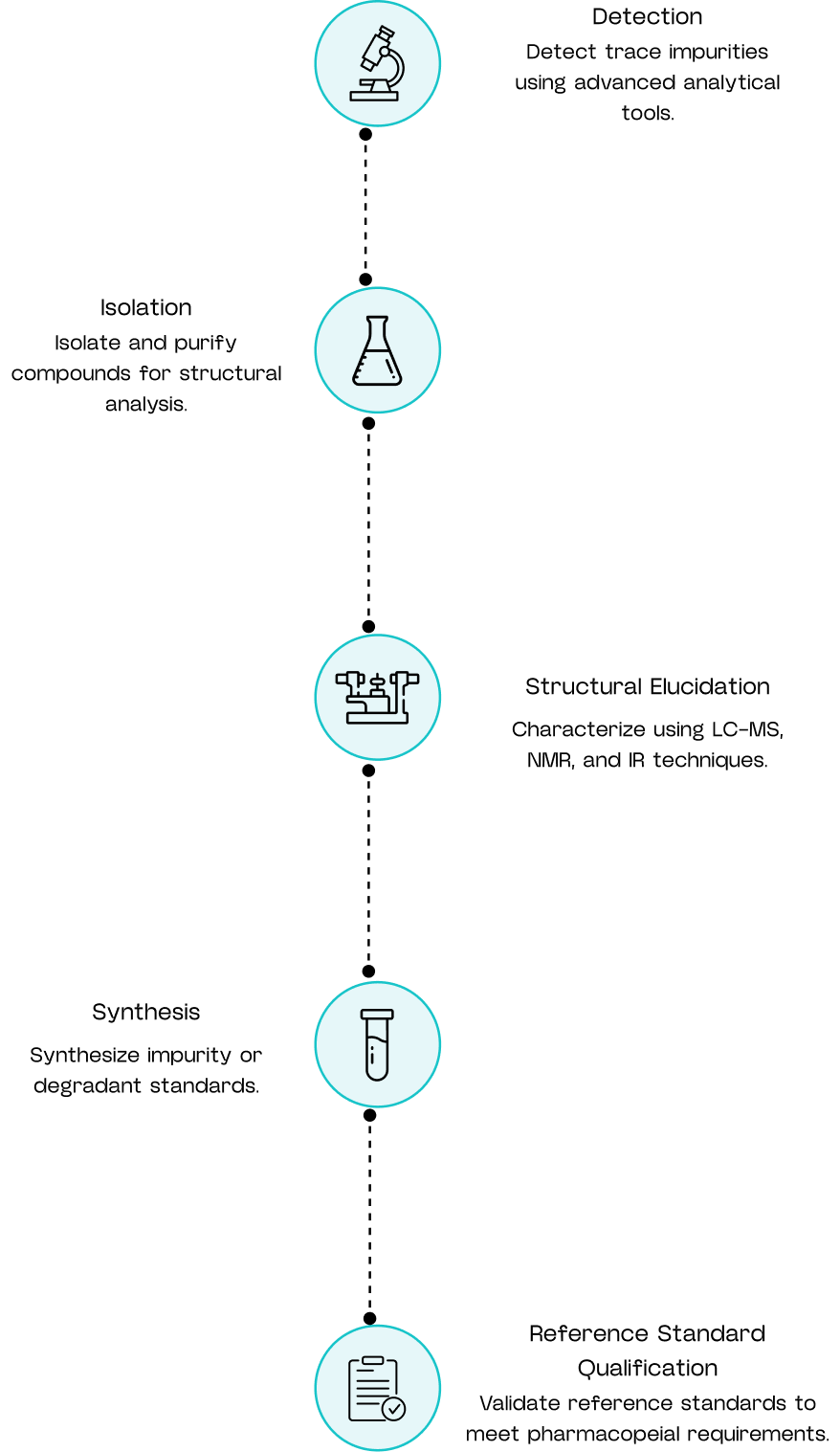

Custom Impurity Synthesis
We synthesize a wide range of process-related and degradation impurities, including:
- Known and unknown process impurities.
- Degradation products from forced stability studies.
- Genotoxic and trace-level impurities.
- Metabolite and structural analog standards.

Each impurity is produced with high purity, full structural confirmation, and batch reproducibility to support analytical and regulatory needs.
Process Optimization & Scale-Up
Our team develops efficient and scalable synthetic routes for impurity standards:
Route design aligned with
the origin ofimpurity formation
Optimization for yield, purity,and reproducibility
Scalable processes to supply milligram
to gram or larger quantities as required
This ensures consistent material availability throughout development, validation, and commercial phases
Analytical Characterization & Impurity Profiling
We provide comprehensive analytical support to confirm identity and purity:
LC–MS, GC–MS, and
HRMS
NMR (¹H, ¹³C, and 2D techniques)
Residual solvent and moisture analysis
Our impurity profiling expertise helps clients understand formation pathways, control strategies, and regulatory documentation requirements.
Regulatory-Ready Reference Standards

Regulatory-Ready Reference Standards
All impurity and reference standards are supplied with:
- Detailed analytical data packages.
- Certificates of Analysis (CoA).
- Documentation suitable for CMC filings and regulatory submissions.
Our materials support method development, validation, stability studies, and quality control testing in line with global regulatory expectations.
Why Choose Elitekem
Analytical Precision
Our impurity profiling is supported by advanced chromatographic and spectroscopic methods for confident identification and quantification.
Tailored Reference Standards
We custom-synthesize impurities and degradation products that match your molecule’s profile and regulatory submission needs.
Regulatory Compliance
Our documentation packages meet ICH guidelines and support ANDA/NDA filings with complete impurity data sets.
Expert Team
Ensure reliable impurity tracking from early R&D through regulatory submission stages.
Your Partner in API Impurity Solutions
From custom impurity synthesis to fully characterized reference standards,
Elitekem Labs helps ensure your API meets the highest standards of quality,
safety, and regulatory compliance.
Your Partner in API Impurity Solutions
From custom impurity synthesis to fully characterized reference standards, Elitekem Labs helps ensure your API meets the highest standards of quality, safety, and regulatory compliance.
